Meet the mitochondria
You’ve heard of the genome – our complete set of DNA – but do you know about mitochondrial DNA and its relevance to health?
Broadly speaking, when we talk about DNA and the genome, we refer to the information packaged into chromosomes and stored in the nucleus of nearly all of an organism’s cells; but mitochondrial DNA is just a little bit different…
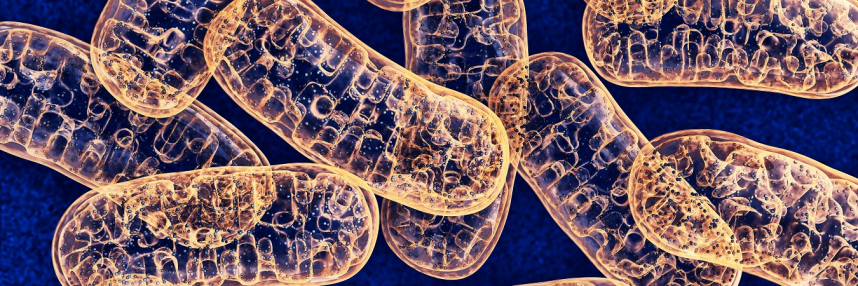
What are mitochondria?
Mitochondria are organelles, small structures within cells that perform specific functions in the cell like the organs do in the body. Mitochondria are found in the cytoplasm of most of the cells in our bodies.
The chemical reactions that provide energy to power the cell’s functions all take place in the mitochondria. Quantities of mitochondria per cell vary between cell types, with tissues that need a lot of energy – such as muscle – having greater numbers.
Crucially, mitochondria have their own genome – 37 genes contained on a circular molecule of DNA. This genome is entirely separate to the genome held in the nucleus of the majority of our cells. Interestingly, the mitochondrial genome was sequenced first, in 1981 – 22 years before the Human Genome Project was completed.
Mitochondrial disease
Mitochondrial disease can result from variation in the mitochondrial genome or from variation in genes in the nuclear genome that are crucial to mitochondrial function.
Leigh syndrome is perhaps the best known of the mitochondrial diseases. The condition usually becomes apparent in a child’s first year and causes a progressive loss of motor abilities, often proving fatal in early childhood; although some individuals experience later onset and slower progression of the disease.
Mitochondrial diseases caused by changes in the nuclear genome include the disorder that affected Charlie Gard: mitochondrial DNA depletion syndrome (MDDS). MDDS is associated with variations in a handful of chromosomal genes, and is inherited in an autosomal recessive pattern, meaning that both parents must be carriers of the same variant.
More broadly speaking, variations in mitochondrial DNA can cause a wide variety of symptoms including seizures, deafness, vision loss and diabetes.
Over 600 of the participants recruited to the 100,000 Genomes Project had suspected mitochondrial disorders, and the researchers have reviewed 365 different genes associated with these conditions, of which 245 are deemed to have sufficient evidence of association to be considered important.
Maternal inheritance
Mitochondria are found outside the cell nucleus in the cytoplasm, which is contributed by the egg rather than the sperm when an embryo is formed. This means that we normally inherit mitochondria only from our mothers, and not our fathers.
The mode of heredity of mitochondrial DNA is complex and unpredictable and does not follow the rules of mendelian genetics. In many cases, children with severe mitochondrial disease are born to seemingly healthy women. The woman may have a mix of healthy and unhealthy mitochondria in her cells. Each time her cells divide – including when her eggs were formed – the mitochondria and cytoplasm are split between two daughter cells. By sheer chance, these daughter cells can sometimes end up with a higher concentration of the unhealthy mitochondria, and, if this is repeated over multiple generations of cell division, an egg may end up containing too small a proportion of the healthy mitochondria.
Scientists have found that some mitochondria can be inherited from the father as well as the mother. Interestingly, the phenomenon of males passing on their mitochondria appears to be governed by an autosomal dominant mutation in the nuclear genome, although the precise gene responsible and its mechanism of action have yet to be identified. Although thought to be rare, it is difficult to estimate how many families might be affected, as very few people ever have their mitochondrial genomes sequenced. Much more research is needed in the area before we can have a clear picture of the mitochondria and its impact on health, and before we can fully appreciate inheritance patterns.
–