Hereditary haemochromatosis
Hereditary haemochromatosis is characterised by an excess of iron in particular organs in the body. It affects both sexes equally, though women often present later than men. It is caused by pathogenic variants in the HFE gene.
Overview
Hereditary haemochromatosis is an autosomal recessive condition characterised by progressive iron overload in the body, leading to multiple organ damage and dysfunction.
Clinical features
Hereditary haemochromatosis (HH) typically presents between 30 to 60 years of age. Although both sexes are equally likely to have the pathogenic genetic variants that cause HH, clinical features are more commonly seen in males. It is common within Northern European populations, especially in those with a Celtic heritage. Women tend to present roughly 10 years later than men, and often after menopause.
Early diagnosis is not easy, as many of the symptoms are common and non-specific. They include:
- weakness and lethargy;
- joint pain;
- weight loss; and
- loss of sex drive.
Given the non-specific nature of the early features, it is vital for clinicians to be alert to family history, particularly because early diagnosis directly improves prognosis.
Later features of HH, which are caused by excessive parenchymal storage of iron in target organs, include:
- liver manifestations, such as:
- abnormal liver blood tests;
- chronic liver disease; and
- hepatocellular carcinoma, which accounts for one third of HH-related deaths;
- arthropathy, affecting:
- second and third metacarpophalangeal joints;
- hips and knees; and
- ankles;
- diabetes mellitus;
- hypogonadism, leading to decreased libido, erectile dysfunction or premature menopause; and
- increased skin pigmentation (bronzing)
Due to the loss of iron during menstruation, women are less likely than men to develop clinical features of iron overload. If HH is diagnosed before the development of cirrhosis or diabetes mellitus, and treated adequately by venesection, life expectancy can be normal. However, if the diagnosis is made after the onset of irreversible organ damage, life expectancy is significantly reduced, primarily due to the risk of liver cancer.
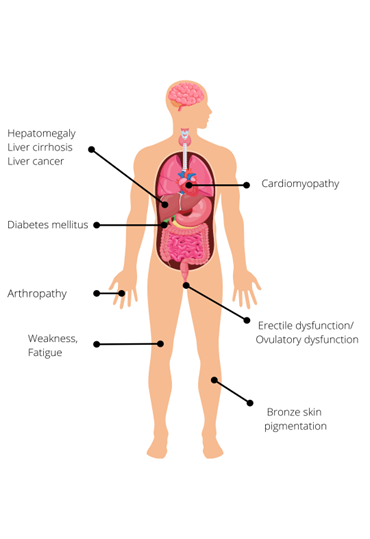
Figure 1: Clinical features of hereditary haemochromatosis.
Genomics
HH is caused by pathogenic variants in the HFE gene. Pathogenic variants in this gene interfere with iron homeostasis, leading to inappropriately high iron absorption by the intestinal mucosa.
The dominant pathogenic variant causing HH is C282Y (p.Cys282Tyr). Around 90% of patients with clinical HH are homozygous for C282Y (both their copies of the HFE gene have this variant), as this genotype is most at risk for iron overload.
Another extremely common HFE variant is pH63D (p.His63Asp). This variant, itself, carries a very low risk although individuals with one C282Y and one H63D pathogenic variant (known as compound heterozygotes) may accumulate low levels of iron, but the risk of developing clinical HH is much lower than that of C282Y homozygotes. H63D homozygotes are also far less likely to have clinical features.

Figure 2: The most common genotypes for hereditary haemochromatosis with reported allele frequencies, where red represents the C282Y variant, blue the H63D variant and green a non-pathogenic variant.
Penetrance for HH is incomplete and the condition shows variable disease expression. This means that many individuals with HH may never manifest serious clinical features. Clinical penetrance (the proportion of individuals who develop clinical features) is much less common than biochemical penetrance (the proportion of individuals with increased transferrin saturation and serum ferritin concentration but no clinical features). For this reason, population screening for the HFE genotype is not recommended.
Non-HFE HH (R96 Iron metabolism disorders – NOT common HFE variants) is much rarer, being a genetically heterogeneous group of iron overload disorders caused by pathogenic variants in hemojuvelin (HJV), hepcidin (HAMP), transferrin receptor 2 (TFR2) and ferroportin (SLC40A1) genes.
Diagnosis
Diagnosis involves the demonstration of iron overload with serum ferritin levels of more than 300µg/L in men, more than 200µg/L in women and transferrin saturations of over 50% in men, over 45% in women.
Occasionally increased liver iron content is identified on liver biopsy or magnetic resonance imaging (MRI).
C282Y homozygosity confirmed via the National Genomic Test Directory‘s ‘R95 Iron overload – hereditary haemochromatosis testing genomic testing’ establishes the diagnosis of HH.
For more information see Presentation: Patient with unexplained iron overload.
Inheritance and genomic counselling
HH is inherited in an autosomal recessive manner. The parents of most affected individuals are carriers for the condition. If both parents are carriers, with each pregnancy there is a:
- 1 in 4 (25%) chance of a child inheriting both gene copies with the pathogenic variant and therefore being affected.
- 1 in 2 (50%) chance of a child inheriting one copy of the gene with the pathogenic variant and one normal copy, and therefore being a healthy carrier themselves; and
- 1 in 4 (25%) chance of a child inheriting both normal copies and being neither affected nor a carrier.
For this reason, it is important to clarify the genetic status of the adult siblings and offspring of affected individuals in order to allow prompt initiation of treatment and preventative measures. Since the condition does not manifest before adulthood, testing children is unlikely to be valuable.
Be aware that juvenile HH is a separate, rarer, more severe condition, with a different genetic basis. Families with HH are not generally at risk of juvenile-onset disease.
Genomic testing for HH is widely available through the 7 regional Genomics Laboratory Hubs. Information regarding testing is available through the National Genomic Test Directory. Support and advice on individual clinical cases can be provided by local gastroenterology, hepatology or haematology services.
Management
Treatment of biochemical iron overload via venesection is indicated for all fit patients, with or without clinical features. The frequency of venesections may be individualised depending on the severity of symptoms, age, comorbidities and tolerability of the procedure.
Diet alone cannot treat HH, so affected individuals need not be advised extreme dietary restriction. Limiting iron fortified food or avoiding excess red meat may be helpful.
For more information see Presentation: Patient with iron overload (suspected hereditary haemochromatosis).
Resources
For clinicians
- GeneReviews: HFE-Related hemochromatosis
- NHS England: National Genomic Test Directory
References:
- European Association for the Study of the Liver. ‘EASL Clinical Practice Guidelines on Haemochromatosis‘. Journal of Hepatology 2022: volume 77, issue 2, pages 479-502. DOI: 10.1016/j.jhep.2022.03.033
- Lucas MR, Atkins JL, Pilling LC and others. ‘HFE genotypes, haemochromatosis diagnosis and clinical outcomes at age 80 years: a prospective cohort study in the UK Biobank‘. BMJ Open 2024: volume 14, issue 3, article number e081926. DOI: 10.1136/bmjopen-2023-081926
- Pilling LC, Tamosauskaite J, Jones G and others. ‘Common conditions associated with hereditary haemochromatosis genetic variants: Cohort study in UK Biobank’. British Medical Journal 2019: volume 364, article number k5222. DOI: 10.1136/bmj.k5222
- Singh P, Mortimore G, Hicks K and others. ‘Venesection Treatment in Haemochromatosis – current best practice from the BSG/BASL Special Interest Group‘. Frontline Gastroenterology 2025: article number 103172. DOI: 10.1136/flgastro-2025-103172
For patients
- British Liver Trust: Haemochromatosis
- Haemochromatosis UK: What is genetic haemochromatosis?
- NHS Health A to Z: Haemochromatosis: Symptoms