Results: Patient with breast cancer and a somatic (tumour) AKT1 variant
The identification of a somatic (tumour) AKT1 variant in a patient with metastatic oestrogen receptor-positive, human epidermal growth factor receptor-2-negative breast cancer who has progressed on endocrine therapy may have implications for the clinical management of the current cancer.
Example clinical scenario
A 58-year-old post-menopausal woman with metastatic oestrogen receptor (ER)-positive, human epidermal growth factor-2 (HER2)-negative breast cancer has been on a cyclin-dependent kinase 4 and 6 (CDK4/6) inhibitor and letrozole for two years and has developed progressive liver metastases. A liver biopsy confirms that the cancer is still ER-positive and HER2-negative. The liver biopsy is submitted for massively parallel sequencing (sometimes called next-generation sequencing) of a large panel of cancer-related genes as a screening investigation for a clinical trial. A somatic (tumour) variant in the AKT1 gene is reported.
Impact of the genomic result
AKT variants in ER-positive, HER2-negative breast cancer
- Endocrine therapy, with or without the use of a CDK4/6 inhibitor, is the standard treatment for patients with ER-positive, HER2-negative metastatic breast cancer; however, most patients will go on to develop treatment resistance.
- The AKT genes (AKT1, 2 and 3) encode the AKT serine-threonine protein kinases (also referred to as protein kinase B alpha, beta and gamma). These proteins are phosphorylated by phosphoinositide 3-kinase (PI3K), and are key components of the signalling pathways which control cell proliferation, survival, metabolism and angiogenesis in both normal and malignant cells.
- Overactivation of the PI3K-AKT-PTEN signalling pathway, as a result of an activating PIK3CA (gene encoding for the p110α subunit of phosphoinositide 3-kinase (PI3K)) or AKT1 variant or an inactivating variant in PTEN, is found in about half of all ER-positive, HER2-negative breast cancers.
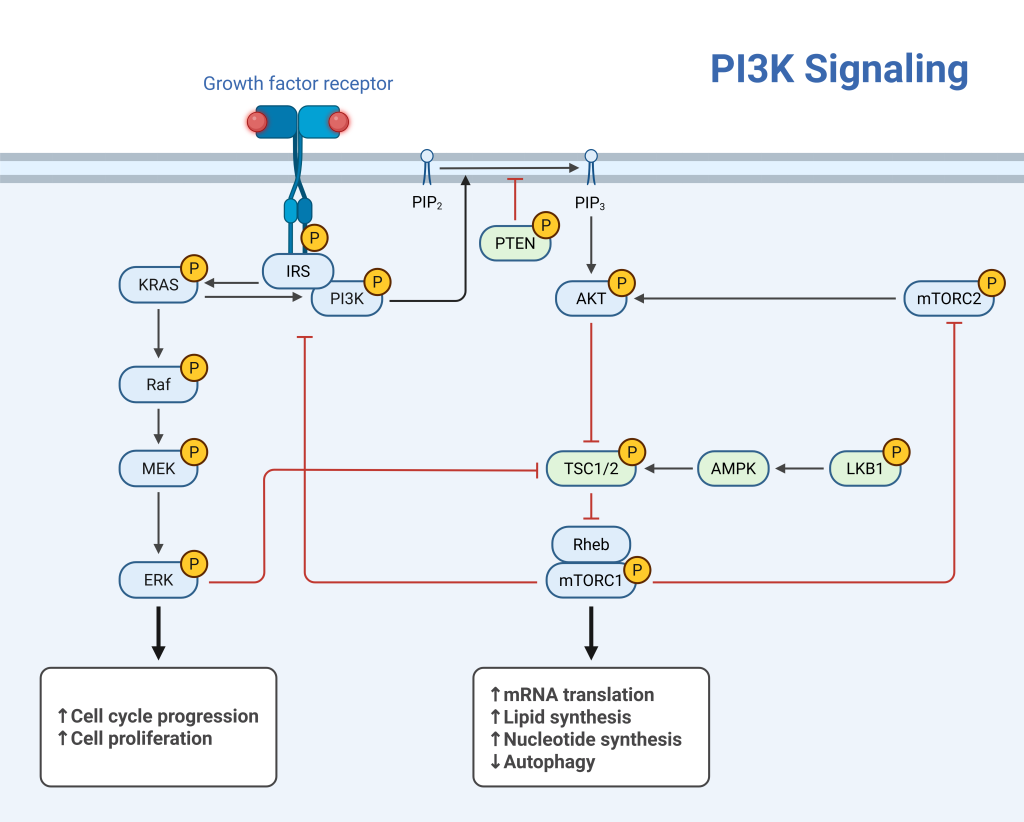
Figure 1: PI3K signaling pathway
Diagram from BioRender (Lugano, G. (2026)
- Capivasertib is a first-in-class small-molecule inhibitor of AKT1, AKT2 and AKT3.
- The phase II randomised CAPItello-291 clinical trial compared treatment with capivasertib plus fulvestrant to placebo plus fulvestrant in patients with ER-positive, HER2-negative advanced breast cancer who had progressed during or after treatment with an aromatase inhibitor (with or without previous CDK4/6 inhibitor therapy). Progression-free survival (PFS) was increased with capivasertib in all patients. The greatest benefit was seen in patients who had a genomic variant in the AKT pathway (defined as activating alteration in PIK3CA or AKT1, or inactivation of PTEN), with a PFS of 7.3 months in the capivasertib-fulvestrant group compared to 3.1 months in the placebo-fulvestrant group (hazard ratio 0.50; 95% CI, 0.38 to 0.65; P<0.001).
What do you need to do?
Management of the current cancer
- Capivasertib, in combination with fulvestrant, has been approved by the US Food and Drug Administration for the treatment of adult patients with hormone receptor (HR)-positive, HER2-negative locally advanced or metastatic breast cancer with one or more biomarker alterations (PIK3CA, AKT1 or PTEN). Eligibility requires progression on at least one endocrine-based regimen in the metastatic setting or recurrence on or within 12 months of completing adjuvant therapy.
- Capivasertib is was approved by the European Medicines Agency in April 2024. There are ongoing discussions with NICE about approval within England.
- Access to novel endocrine therapies for metastatic breast cancer with AKT variants may be available in the context of clinical trials.
- AKT1 variants are almost exclusively somatic, and common in breast cancers, such that the finding of such a variant in tumour-derived DNA would not routinely trigger any onward constitutional (germline) genomic testing of patients with breast cancer in the absence of syndromic features. Constitutional (germline) pathogenic variants in AKT1 have been reported in exceptionally rare patients with features suggestive of Cowden syndrome (PTEN hamartoma tumour syndrome). AKT1 variants are almost exclusively somatic, and common in breast cancers, such that the finding of such a variant in tumour-derived DNA would not routinely trigger any onward constitutional (germline) genomic testing of patients with breast cancer in the absence of syndromic features. Patients with syndromic features (e.g. macrocephaly, mucocutaneous features) should be referred to clinical genetics for an assessment.
- For information about how to arrange testing in Wales, Scotland or Northern Ireland, see Genomic testing in the devolved nations.
- If you are discussing genomics concepts with your patients, you may find it helpful to use the visual communication aids for genomics conversations.
Resources
For clinicians
References:
- Gennari A, André F, Barrios CH and others. ‘ESMO clinical practice guideline for the diagnosis, staging and treatment of patients with metastatic breast cancer‘. Annals of Oncology 2021: volume 32, issue 12, pages 1,475–1,495. DOI: 10.1016/j.annonc.2021.09.019
- Turner NC, Oliveira M, Howell SJ and others for the CAPItello study group: ‘Capivasertib in hormone receptor-positive advanced breast cancer‘. The New England Journal of Medicine 2023: volume 388, issue 22, pages 2,058–2,070. DOI: 10.1056/NEJMoa2214131